CUHK
News Centre
CUHK Discovers New Inhibitors to Restrain NeurodegenerationShedding Light on Treating PolyQ Diseases
Professor H.Y. Edwin Chan, Professor of the School of Life Sciences at The Chinese University of Hong Kong (CUHK) and his research team have discovered a small molecule compound termed Anti-polyQ Aggregation for Machado-Joseph-Associated Neurodegeneration (AQAMAN) which can dissociate toxic protein aggregates and restrain neurodegeneration. AQAMAN has the potential to be developed into therapeutic usage, giving hope to individuals who suffer from Machado-Joseph Disease, the most common form of spinocerebellar ataxias (SCAs), as well as other polyglutamine (polyQ) diseases. Professor Chan’s research findings have been recently published in the Journal of Biological Chemistry as a cover story.
Since 1999, Professor Chan has been studying polyQ diseases, a group of rare neurological disorders including SCAs, and developing therapeutic interventions against these diseases. SCAs cause progressive death of nerve cells in the cerebellum of the human brain, leading to failure of muscle control in the arms and legs, lack of balance and coordination of gait and slurred speech. In general, polyQ disease patients will gradually lose the fine motor functions of their bodies and have difficulty maintaining balance or coordinating daily movements. To date, polyQ diseases remain incurable.
The discovery of AQAMAN in treating polyQ diseases
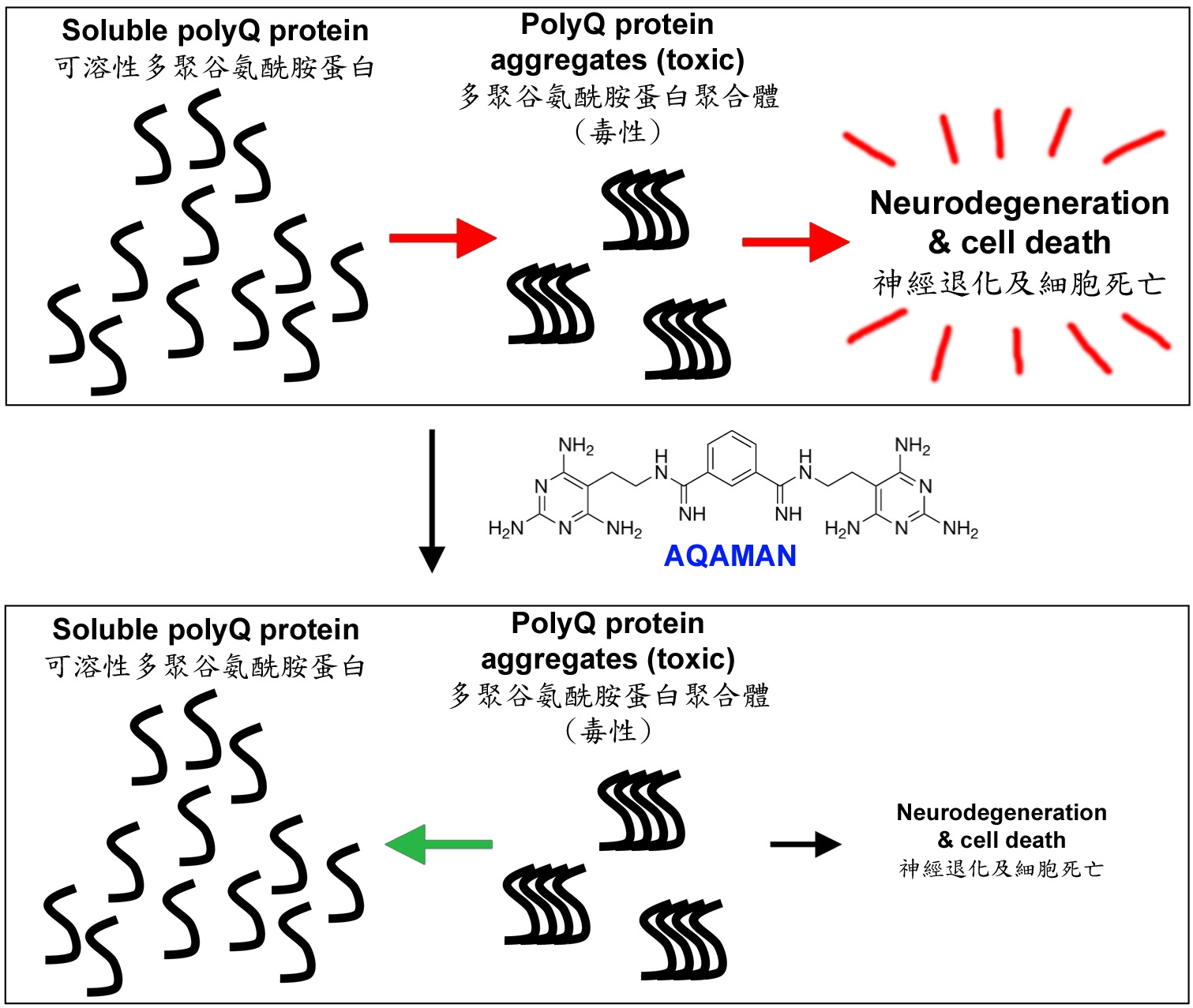
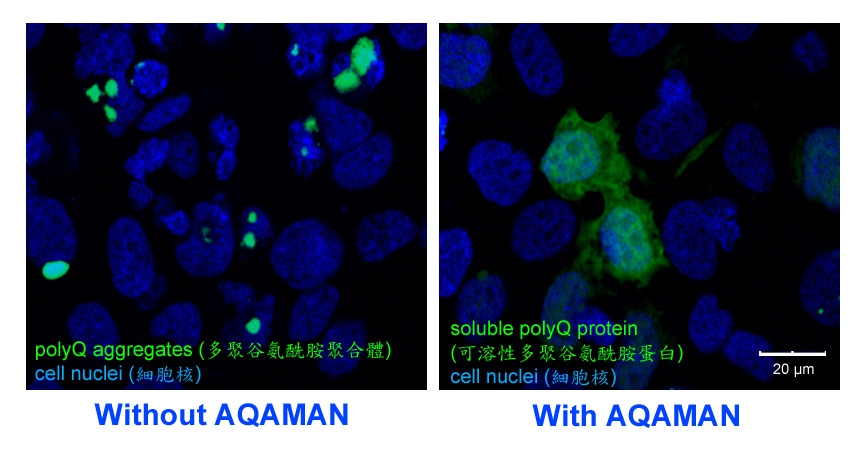
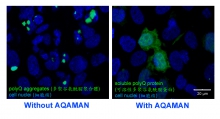
Professor Chan established an international research collaboration on SCAs and other rare neurological and neuromuscular diseases, and co-manages multi-disciplinary research programmes with his collaborators in Denmark, France, Italy, the UK and the US. In particular, Professor Chan works closely with Professor Steven C. Zimmerman (University of Illinois at Urbana-Champaign, US), an expert in synthetic chemistry, to screen for drug candidates that target rare diseases including SCAs and muscular dystrophies. The AQAMAN small molecule compound was synthesised by the Zimmerman group and was originally designed to target muscular dystrophies. Professor Chan’s team serendipitously demonstrated translational potentials of AQAMAN in treating polyQ diseases, a group of neurodegenerative disorders that are characterised as protein aggregation diseases. AQAMAN can interfere with toxic polyQ protein aggregate formation as well as to promote its deaggregation. Professor Chan’s team is currently seeking pharmaceutical collaboration partners to further improve the drug-likeness of AQAMAN.
Professor Chan said, “Protein misfolding and aggregation are known to associate closely with many human diseases. Since 1999, I have been studying how polyQ protein aggregation causes nerve cell degeneration and developing genetic strategies to manipulate toxic polyQ aggregate formation. In 2012, I initiated a collaboration with Professor Zimmerman to use the fruit fly Drosophila as a drug screening model to evaluate small molecule compounds for muscular dystrophies. Later in 2016, I teamed up with Professor Alex C. Koon (School of Life Sciences, CUHK) who studies disease mechanisms of muscular dystrophies using the fruit fly neuromuscular junction. AQAMAN is among the many bisamidine-based inhibitors that were designed to inhibit RNA-protein complex in muscular dystrophies that we are studying. It was totally out of our expectation that AQAMAN has the ability to interfere with toxic protein aggregation. We hope AQAMAN can further be developed into therapeutic usage and bring hope to the sufferers of polyQ disorders at the bed-side.”
This work was supported by the CUHK Vice-Chancellor’s One-Off Discretionary Fund, the CUHK Lui Che Woo Institute of Innovative Medicine, the CUHK Gerald Choa Neuroscience Centre, The Croucher Foundation, the National Institutes of Health (US), and donations from the Hong Kong Spinocerebellar Ataxia Association.
An improved peptidylic inhibitor against RNA toxicity of neurodegenerative diseases
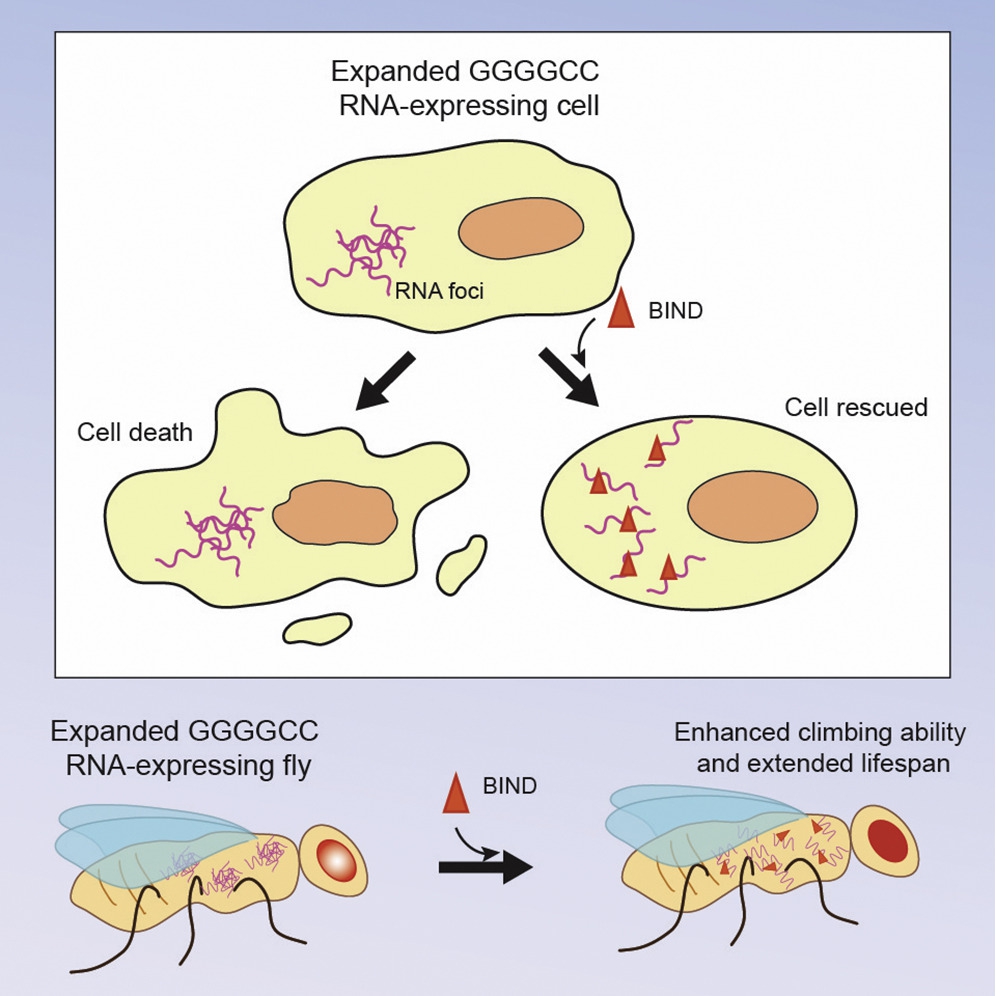
In 2016, Professor Chan teamed up with Professor Jacky C.K. Ngo (School of Life Sciences, CUHK), a structural biologist experienced in drug discovery, to develop a novel peptidylic inhibitor against CAG RNA toxicity of polyQ diseases. This collaborative team has recently reported an improved version of peptidylic inhibitor termed beta-structured inhibitor for neurodegenerative diseases (BIND). It is a synthetic peptide with a length of 21 amino acids, and has the ability to target RNA toxicity of polyQ diseases and C9orf72-associated amyotrophic lateral sclerosis/frontotemporal dementia (C9ALS/FTD). The findings on polyQ and C9ALS/FTD have been published in RNA (Zhang et al., (2018)) and Molecular Therapy – Nucleic Acids (Zhang et al., (2019) in press) respectively.
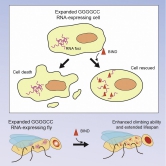
Professor Ngo said, “Since 2016, Professor Chan and I have worked very hard to further optimise our peptidylic RNA toxicity inhibitors. RNA toxicity in polyQ is caused by CAG and in C9ALS/FTD by GGGGCC repeat expansion. GGGGCC is a hexanucleotide repeat DNA sequence found in the human genome. Uncontrolled expansion of the GGGGCC repeat causes the rare disease C9ALS/FTD. Although polyQ and C9ALS/FTD have distinct disease mechanisms of RNA toxicity, our biophysical studies clearly show that BIND can inhibit both kinds of toxicity and mitigate neurodegeneration in both types of diseases. We plan to further optimise the stability and brain-targeting property of BIND and aim at bringing our study to the pre-clinical stage.”
February 28 is Rare Disease Day. Rare Disease Day is a worldwide campaign first organised in 2008 with the aim of raising awareness amongst the general public and decision-makers of rare diseases and their impact on patients’ lives. The Hong Kong Spinocerebellar Ataxia Association was established in 2007 and the Hong Kong Alliance for Rare Diseases in 2015 to provide support to patients and their families and promote, in general, rare diseases awareness in our community.
Brief biography of Professor Edwin H.Y. Chan
Professor H.Y. Edwin Chan is Professor in the School of Life Sciences, CUHK. He received undergraduate training in biochemistry from CUHK, doctoral training at The University of Cambridge (UK), and postdoctoral training at The University of Pennsylvania (US). Since 1999, Professor Chan has been investigating the pathogenic pathways of rare neuronal diseases. His contribution to this field has been recognised by academia through the various research prizes he has received, including the CUHK Young Researcher Award and the Genetics Society of China 13th Ju-Chi Li Animal Genetics Prize. Professor Chan is a Founding Member and Executive Committee member of The Young Academy of Sciences of Hong Kong. He is an advisor to the Hong Kong Spinocerebellar Ataxia Association and the Hong Kong Alliance for Rare Diseases.
Brief biography of Professor Alex C. Koon
Professor Alex C. Koon is Research Assistant Professor and Associate Director of the Laboratory of Drosophila Research in the School of Life Sciences, CUHK. He received undergraduate training in biochemistry from Boston College (US), doctoral training at The University of Massachusetts Medical School (US), and postdoctoral training at The University of Queensland (Australia). Professor Koon studies synaptic plasticity and developmental neurobiology using the fruit fly neuromuscular junction. Since 2016, his research interests have expanded to include the underlying mechanisms of neuromuscular diseases, such as myotonic dystrophies. He is an active member of the Society for Neuroscience (US).
Brief biography of Professor Jacky C.K. Ngo
Professor Jacky C.K. Ngo is Assistant Professor in the School of Life Sciences, CUHK. He received undergraduate and doctoral training in chemistry and biochemistry from the University of California San Diego (US), and postdoctoral training at the Beth Israel Deaconess Medical Center-Harvard Medical School (US). Professor Ngo studies the structure-function relationship of proteins involved in the regulation of pre-mRNA splicing. Since 2011, his research interests have expanded to include target-based discovery and development of inhibitors for molecular targets in cancers and neurodegenerative diseases. He is an active member of the RNA Society (US) and American Society of Biochemistry and Molecular Biology (US).
JBC cover story: AQAMAN is displayed as a hero breaking down toxic protein aggregations inside a cell.
AQAMAN can interfere with toxic polyQ protein aggregate formation as well as to promote its deaggregation.
BIND is a potent peptidylic inhibitor that suppresses expanded GGGGCC RNA-mediated neurodegeneration, enhances climbing ability and extends the lifespan of disease flies.